Reference | 化學工廠資訊網
2009年4月1日—TheUSPharmacopeia(USP)definesreference-standardmaterialsashighlycharacterizedspecimensofdrugsubstances,excipients,reportable ...
 The user (custom manufactures or synthesizes the reference
standard)
Companies such as chemical suppliers.
The user (custom manufactures or synthesizes the reference
standard)
Companies such as chemical suppliers.
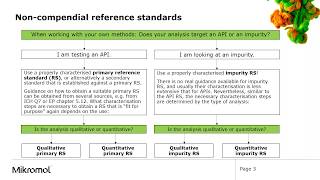
Reference-standard materials that are synthesized by the user or supplied by a contract manufacturer or secondary company must be characterized (3). Both the reference standards and drug substance may be synthesized initially using the same process. The reference standard should be of the highest purity possible; the drug substance may require further purification to become a reference standard (additional purification steps used for a drug substance should be fully described and included in any regulatory filing).
Storage and impurity detection
Impurities classified as organic (process and drug related), inorganic, or residual solvents (4) can be introduced during the manufacturing process for the drug substance, drug product, or excipient and/or through stor...
USP Reference Standards | 化學工廠資訊網
reference standard | 化學工廠資訊網
All Reference Standards | 化學工廠資訊網
Drug reference standard | 化學工廠資訊網
Reference standards | 化學工廠資訊網
Reference | 化學工廠資訊網

【臺南市】台灣標準品股份有限公司
臺南市化工廠有哪些?本篇為大家整理位於安南區鹽田里工業二路31號的「台灣標準品股份有限公司」相關資訊,用列表的方式整理...